Principle: Here alcohol group of benzoin is oxidized to ketone group forming benzil in the presence of concentrated nitric acid. Nitration of aromatic ring is not occurring as sulphuric acid is totally absent in the whole process.
Which is the best oxidising agent used for synthesis of benzil from benzoin?
H2O2 is inexpensive and environmental friendly oxidant. It is observed that the homogeneous catalyst is very active in high conversion rate of benzoin to benzil.
What happens when you add too much water when attempting to dissolve the thiamine hydrochloride?
Too much water will force benzaldehyde out of solution preventing an efficient reaction. Too little water prevents the thiamine hydrochloride from dissolving. Some of the base reacts with the thiamine hydrochloride to produce thiamine which is the active catalyst.
How is benzil produced?
Preparation. Benzil is prepared from benzoin, for example with copper(II) acetate: PhC(O)CH(OH)Ph + 2 Cu2+ → PhC(O)C(O)Ph + 2 H+ + 2 Cu. Other suitable oxidizing agents such as nitric acid (HNO3) are used routinely.What is the role of nitric acid in synthesis of benzil?
Principle: Here alcohol group of benzoin is oxidized to ketone group forming benzil in the presence of concentrated nitric acid. Nitration of aromatic ring is not occurring as sulphuric acid is totally absent in the whole process.
What is the chemical formula for nitric acid?
A nitrogen oxoacid of formula HNO3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen atoms. Nitric acid (HNO3), also known as aqua fortis (Latin for “strong water”) and spirit of niter, is a highly corrosive mineral acid.
What is benzil used for?
Benzil is used as a pharmaceutical intermediate and uv curing resin photosensitizer. In polymer chemistry, it is used as a photoinitiator. Further, it serves as a potent inhibitor of human carboxylesterases. It is used in the preparation of diketimines by reacting with amines.
What is Iupac name of Benzil?
IUPAC Name1,2-diphenylethane-1,2-dioneMolar Mass210.232 g/molInChIInChI=1S/C14H10O2/c15-13(11-7-3-1-4-8-11)14(16)12-9-5-2-6-10-12/h1-10HInChI KeyWURBFLDFSFBTLW-UHFFFAOYSA-NCAS Number 134-81-6Is Benzil an acid or base?
The rearrangement of benzil is base (and not acid) catalyzed under conventional conditions (water–dioxane mixture around 100 °C).
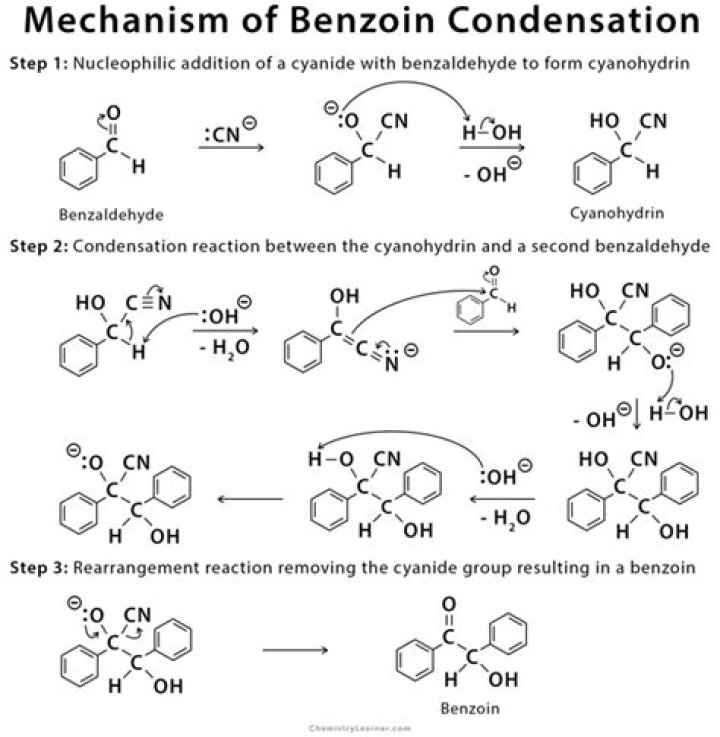
Is benzoin a ketone?Benzoin is a ketone that consists of acetophenone bearing hydroxy and phenyl substituents at the alpha-position. … It is a member of benzoins and a secondary alpha-hydroxy ketone. ChEBI. Benzoin is a white crystalline compound prepared by condensation of benzaldehyde in potassium cyanide, and is used in organic syntheses …
Article first time published onWhat is the role of cyanide in benzoin condensation reaction?
The reaction mechanism was proposed in 1903 by A. J. Lapworth. In the first step in this reaction, the cyanide anion (as sodium cyanide) reacts with the aldehyde in a nucleophilic addition. … Proton transfer and elimination of the cyanide ion affords benzoin as the product.
What is the role of thiamine hydrochloride in the synthesis of benzoin?
The objective of this experiment is to synthesize benzoin from the benzaldehyde, using thiamine hydrochloride. … NaOH attacks thiamine and deprotonates it. It is a condensation, since a molecule of H2O is released. There is a formation of a carbine, which is very reactive.
What is the role of the hydroxide reagent in the thiamine catalyzed benzoin condensation?
What is the purpose of the sodium hydroxide solution? the sodium hydroxide solution deprotonates the thiamine salt. Thiamine hydrochloride contains very acidic proton located near the positively charged nitrogen atom. The strong base removes this proton, thus forming thiamine in its free form.
Why is it important to use fresh benzaldehyde?
Why is it important to use fresh benzaldehyde? The solubility of thiamine hydrochloride is much higher in water than in ethanol. Thus, it is easier to dissolve the thiamine salt in water and add 95% ethanol afterwards. … In other words, the sodium hydroxide allows the thiamine catalyst to be formed in-situ.
What is Benzil Benzilic acid rearrangement?
The benzilic acid rearrangement is formally the 1,2-rearrangement of 1,2-diketones to form α-hydroxy–carboxylic acids using a base. This reaction receives its name from the reaction of benzil with potassium hydroxide to form benzilic acid. … The reaction is formally a ring contraction when used on cyclic diketones.
What is the function of resorcinol?
Resorcinol is used in acne and skin treatment medication to remove hard scaly or rough skin. The concentration in acne medicine is usually about two percent. When resorcinol is used as a dermatological treatment, it is likely to be applied to damaged skin, which allows more resorcinol to enter the body.
What is the meaning of Benzil?
Definition of benzil : a yellow crystalline diketone C6H5COCOC6H5 made by oxidizing benzoin.
What functional group is Benzil?
Benzil is an alpha-diketone that is ethane-1,2-dione substituted by phenyl groups at positions 1 and 2 respectively. It is an alpha-diketone and an aromatic ketone.
Why is nitric acid HNO3?
Nitric acid is a highly corrosive acidic substance. … Its chemical formula is HNO3, so it has one hydrogen (H) atom, one nitrogen (N) atom, and three oxygen (O) atoms. All three oxygen (O) atoms are bonded to the nitrogen (N) atom. The nitrogen atom carries a charge of +1, and one oxygen atom carries a charge of -1.
What is the use of nitric acid?
Nitric acid is used for the production of ammonium nitrate, a major component of fertilizers. It is also used for producing explosives like nitroglycerin and trinitrotoluene (TNT) and for oxidizing metals.
Why is nitric acid referred to as an oxidizing acid?
In the case of Nitric acid (HNO3), the nitrogen has a +5 oxidation state, which is its highest oxidation state, so it will get reduced and oxidize other molecules. Hence it acts as an oxidizing agent.
Does Benzil give tollens test?
Even 1,2-diketones like Dialkyl Glyoxals, Benzil, Furil, etc. again give this test, due to Benzil-Benzilic Acid Rearrangement followed by S.E.T. Yes, all alpha hydroxy ketones gives tollens test.
Is Benzil toxic?
Skin: Causes skin irritation. Ingestion: May cause irritation of the digestive tract. May cause gastrointestinal irritation with nausea, vomiting and diarrhea. Inhalation: May cause respiratory tract irritation.
Is benzoin more polar than Benzil?
Since benzil is less polar than benzoin, a polar solvent is used as solvent for the recrystallization.
What Colour is benzoin?
It consists of hard, usually cream-coloured/pale orange pieces, which if broken reveal a milky white colour. The benzoin is quite pale in colour when freshly collected but darkens gradually during storage to a sandy-orange colour.
Is benzoin a alpha hydroxy ketone?
Benzoin is an alpha-hydroxy ketone. Ketone having a hydroxyl group at beta-carbon is known as beta-hydroxy ketone.
What is a benzoin condensation reaction?
The Benzoin Condensation is a coupling reaction between two aldehydes that allows the preparation of α-hydroxyketones. The first methods were only suitable for the conversion of aromatic aldehydes.
Which catalyst is used in benzoin condensation?
A classic example is the benzoin condensation, first reported by Wöhler and Liebig in 1832 with a proposed mechanism in 1903 by Lapworth; cyanide is used as a catalyst to effect the dimerization of two benzaldehyde units [13].
What is the purpose of aldol condensation?
Aldol condensations are important in organic synthesis, because they provide a good way to form carbon–carbon bonds. For example, the Robinson annulation reaction sequence features an aldol condensation; the Wieland-Miescher ketone product is an important starting material for many organic syntheses.
Is thiamine a base or acid catalyst?
As a base it can form salts with acids, such as hydrochloride. It is stable at acidic pH, but is unstable in alkaline solutions. Thiamine, which is a persistent carbene, is used by enzymes to catalyze benzoin condensations in vivo.
Why thiamine catalyzed benzoin condensation is called green synthesis?
reaction is rapid conversion, solvent free; no use of catalyst and hazardous chemicals in the synthesis hence, it is ecofriendly green chemistry synthesis.