volatile. noun. Definition of volatile (Entry 2 of 2) : a substance that is readily vaporizable at relatively low temperature : a volatile substance.
What is volatile and non volatile in chemistry?
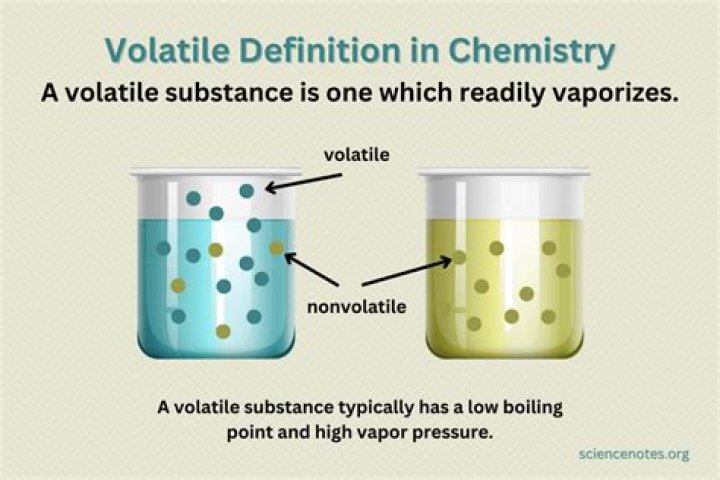
A non-volatile substance refers to a substance that does not readily evaporate into a gas under existing conditions. … A volatile substance is one that evaporates or sublimates at room temperature or below. Volatile substances have higher vapor pressures versus non-volatile substances at the same temperature.
What is example of volatile?
Volatile is defined as a substance that quickly evaporates. An example of volatile is acetone. The definition of volatile is tending to become violent or something that is rapidly evaporating. An example of volatile is a young man who often gets into fist fights.
What is the definition of volatility in chemistry?
3.7 Volatility and Flammability. The volatility of an organic chemical (typically referring to a liquid or liquefied gas) may be defined as its tendency to vaporize, that is, to change from the liquid to the vapor or gaseous state.What is meant by a volatile liquid?
Definition. A substance capable of readily changing from a solid or liquid form to a vapour; having a high vapour pressure and a low boiling point.
What are volatile substances for Class 8?
Volatile SubstancesNon-Volatile Substances8. Examples- Alcohol, Mercury, and Gasoline.8. Examples- Sugar, Oil and salt.
What are volatile substances Class 9?
1 Answers. A volatile substance is is the one which can easily evaporate at room temperature. In general, any substance with a boiling point below 100 degrees Celsius is considered volatile, while all other liquids are called nonvolatile. For example, methanol, acetonitrile, etc are volatile substances.
Which is volatile substance of following?
Volatile compoundCAS Registry NumberButane, iso-butane, propane, liquefied petroleum gas (LPG)106-97-8Chloroform, trichloromethane67-66-3Diethyl ether, ethyl ether, ether, 1,1′-Oxybisethane60-29-7Dimethylether, Methyl Ether, Oxybismethane, DME115-10-6Are gases volatile?
Substances that are gases at room temperature are extremely volatile: they have high volatility. They can only be seen as liquids when exposed to low temperatures or high pressures.
What increases volatility chemistry?Increasing the temperature increases the amount of vapor that is formed and thus the vapor pressure. In a mixture, each substance contributes to the overall vapor pressure of the mixture, with more volatile compounds making a larger contribution.
Article first time published onWhat is volatile impurities in chemistry?
Abstract. Organic volatile impurities are residual solvents that are used in and are produced during the synthesis of drug substances, or in excipients used in the production of drug formulations.
What is another word for volatility?
capriciousnessexcitabilityflightinessmutabilityfitfulnessuncertaintyfluidityoscillationvacillationtransience
What are volatile metals?
Volatile metals like Hg,Zn and Cd are easily purified by distillation as they have low boiling points. The impure metal is heated in a retort and vapours of volatile metals are collected and condensed in a receiver leaving behind non volatile impurities in retort.
Why is alcohol volatile?
Volatility is greater if vapour pressure is greater. Surface molecules behave differently than the molecules in bulk.So evaporation takes place at all temperatures. Alcohols have high vapour pressure hence are highly volatile.
Is ethanol volatile?
Ethanol is a clear, colourless liquid with a characteristic pleasant odour and burning taste. It is highly flammable. Ethanol is used to dissolve other chemical substances and mixes readily with water and many organic liquids. Ethanol is considered a volatile organic compound by the National Pollutant Inventory.
Is water a volatile?
Water (H2O) is moderately volatile. It has a boiling point of 100oC and evaporates only slowly at room temperature. It is not flammable or explosive.
What is volatile and nonvolatile in chemistry class 12?
Volatile substances have a tendency to vaporize whereas nonvolatile substances do not have a tendency to vaporize. Volatile substances have a high vapor pressure at normal room temperature and pressure.
Is alcohol a volatile liquid?
Many organic compounds are volatile. For example, alcohol is volatile. Because volatile substances readily vaporize, they mix with air and can be smelled (if they have an odor).
Is oxygen volatile?
Is oxygen a volatile or refractory element, and why? Pure oxygen is a gas at 1-atmosphere pressure and room temperature. It boils at 90 K (much lower temperature than room temperature, 300 K), and so it is very volatile.
Is oxygen is a volatile substance?
Of the main components of air, oxygen has the highest boiling point and therefore is less volatile than nitrogen and argon. The process takes advantage of the fact that when a compressed gas is allowed to expand, it cools.
What is an example of a VOC?
Volatile Organic Compounds in Your Home. … Common examples of VOCs that may be present in our daily lives are: benzene, ethylene glycol, formaldehyde, methylene chloride, tetrachloroethylene, toluene, xylene, and 1,3-butadiene.
What are volatile solvents?
Volatile solvents are liquids that easily vaporize into a gas. When inhaled, the gas can cause a state of intoxication and long-term nerve and organ damage. Volatile solvents are found in many common household products, such as adhesives, paint, and cleaning fluid.
Is Naphthalene a volatile substance?
Naphthalene is highly volatile and has a characteristic odour; it has been used as moth repellent. In its chemical behaviour, naphthalene shows the aromatic character associated with benzene and its simple derivatives.
Are volatile liquids flammable?
Volatility and Flammability Since it is a liquid’s vapor rather than the liquid itself that ignites when mixed with air, volatile compounds combust more readily and spontaneously than less volatile compounds.
Which compound is most volatile?
Volatility is tendency of a substance to evaporate at normal temperatures. Here 2, 2-dimethyl propane has the least surface area. Thus, van der Waals force of attraction between the molecules is less and hence, lesser amount of energy is required to move them apart. So, it is the most volatile compound among these.
What are the volatile acids?
Volatile acidity refers to the steam distillable acids present in wine, primarily acetic acid but also lactic, formic, butyric, and propionic acids. Commonly, these acids are measured by Cash Still, though now they can be measured by gas chromatography, HPLC or enzymatic methods.
What is non volatile substance?
Nonvolatile is the inability of a substance to readily evaporate into gas. Nonvolatile substances are usually solids with boiling points above 212°F (100°C), and do not vaporize easily at room temperature and pressure.
What is the opposite of volatility?
Antonyms & Near Antonyms for volatility. levelheadedness, practicality, reasonability, reasonableness.
Is co2 volatile?
Volatiles include nitrogen, carbon dioxide, ammonia, hydrogen, methane, sulfur dioxide, water and others.
When a person is volatile?
A person who is volatile loses his or her temper suddenly and violently. A volatile political situation could erupt into civil war. When the stock market is volatile, it fluctuates greatly. And in scientific language, a volatile oil evaporates quickly. Definitions of volatile.
Is mercury volatile metal?
13.28. Metallic mercury, however, is very volatile and vaporizes readily.