Nuclear radiation refers to the particles and photons emitted during reactions that involve the nucleus of an atom. … Nuclear radiation includes gamma rays, x-rays, and the more energetic portion of the electromagnetic spectrum.
Why is it called nuclear radiation?
These forces work toward a strong, stable balance by getting rid of excess atomic energy (radioactivity). In that process, unstable radioactive nuclei may emit energy, and this spontaneous emission is called nuclear radiation.
What is nuclear equation?
Nuclear equations represent the reactants and products in radioactive decay, nuclear fission, or nuclear fusion. Instead of chemical equations where it shows the different number of elements is conserved in a reaction, in a nuclear reaction the atomic mass and proton number are conserved.
What is radiation in physics easy definition?
Radiation is energy that comes from a source and travels through space and may be able to penetrate various materials. Light, radio, and microwaves are types of radiation that are called nonionizing. … Gamma radiation and x rays are examples of electromagnetic radiation.What are three types of radiation?
The three most common types of radiation are alpha particles, beta particles, and gamma rays.
What is radiation examples?
Radiation Examples ultraviolet light from the sun. heat from a stove burner. visible light from a candle. x-rays from an x-ray machine. alpha particles emitted from the radioactive decay of uranium.
What is radiation definition and example?
Radiation is the process of sending off energy in the form of light, heat, x-rays or nuclear particles. An example of radiation are the energy waves off of a nuclear bomb. … The transfer of energy via radiation (as opposed to convection or conduction).
What are the 4 types of nuclear reactions?
- Fission.
- Fusion.
- Nuclear Decay.
- Transmutation.
What is difference between wave and radiation?
Water waves transmit energy through space by the periodic oscillation of matter (the water). In contrast, energy that is transmitted, or radiated, through space in the form of periodic oscillations of electric and magnetic fields is known as electromagnetic radiation.
What is the proton symbol?ParticleSymbolChargeelectrone–1protonp++1neutronno0
Article first time published onWhat is the chemical symbol for uranium?
Uranium is a silvery-white metallic chemical element in the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons.
What is norm and Tenorm?
NORM denotes “Naturally Occurring Radioactive Material” and TENORM stands for Technologically-Enhanced Naturally Occurring Radioactive Material.” NORM and TENORM are important and complex issues that involve science, politics, business, and the public.
Who discovered radiation?
March 1, 1896: Henri Becquerel Discovers Radioactivity. In one of the most well-known accidental discoveries in the history of physics, on an overcast day in March 1896, French physicist Henri Becquerel opened a drawer and discovered spontaneous radioactivity.
What radiation is an electron?
Beta radiation takes the form of either an electron or a positron (a particle with the size and mass of an electron, but with a positive charge) being emitted from an atom.
Is all radiation light?
Sometimes we use the term ‘radiation’ when we mean ‘light’, and vice versa. … It can also be described as a flow of particle-like ‘wave-packets’, called photons, that travel constantly at the speed of light (about 300 000 kilometres per second). Radiation, electromagnetic waves and photons are simply ‘light‘.
What is the definition of radiation for Class 7?
Radiation: The process of the transfer of the heat from one place to another place without heating the intervening medium is called radiation. For example, the heat from the sun reaches the earth through radiation mode.
What is conduction in simple words?
Definition of conduction 1 : the act of conducting or conveying. 2a : transmission through or by means of a conductor also : the transfer of heat through matter by communication of kinetic energy from particle to particle with no net displacement of the particles — compare convection, radiation.
What are the 4 types of radiation?
There are four major types of radiation: alpha, beta, neutrons, and electromagnetic waves such as gamma rays. They differ in mass, energy and how deeply they penetrate people and objects. The first is an alpha particle.
Is radiation a wave or a particle?
In physics, radiation is the emission or transmission of energy in the form of waves or particles through space or through a material medium. This includes: electromagnetic radiation, such as radio waves, microwaves, infrared, visible light, ultraviolet, x-rays, and gamma radiation (γ)
What are the 7 types of radiation?
The EM spectrum is generally divided into seven regions, in order of decreasing wavelength and increasing energy and frequency. The common designations are: radio waves, microwaves, infrared (IR), visible light, ultraviolet (UV), X-rays and gamma rays.
Does sun produce gamma rays?
Our Sun emits light at progressively shorter wavelengths, too: the ultraviolet, X-ray, and even gamma-ray parts of the spectrum. … So, the only gamma rays from the Sun we receive here on Earth are from extreme solar events, such as the most powerful solar flares.
What is the amplitude of radiation?
In electromagnetic waves, the amplitude is the maximum field strength of the electric and magnetic fields. (See Figure 1.) Thus the energy carried and the intensity I of an electromagnetic wave is proportional to E2 and B2.
What is meant by infrared rays?
Infrared radiation (IR), or infrared light, is a type of radiant energy that’s invisible to human eyes but that we can feel as heat. … From highest to lowest frequency, electromagnetic radiation includes gamma-rays, X-rays, ultraviolet radiation, visible light, infrared radiation, microwaves and radio waves.
What are the 3 nuclear particles?
After several years of study, scientists identified several distinct types of particles resulting from radioactive processes (radiation). The three distinct types of radiation were named after the first three letters of the Greek alphabet: (alpha), (beta), and (gamma).
What are the 5 types of nuclear reactions?
- Classes of Radioactive Nuclei.
- Nuclear Decay Reactions.
- Alpha Decay.
- Beta Decay.
- Positron Emission.
- Electron Capture.
- Gamma Emission.
- Spontaneous Fission.
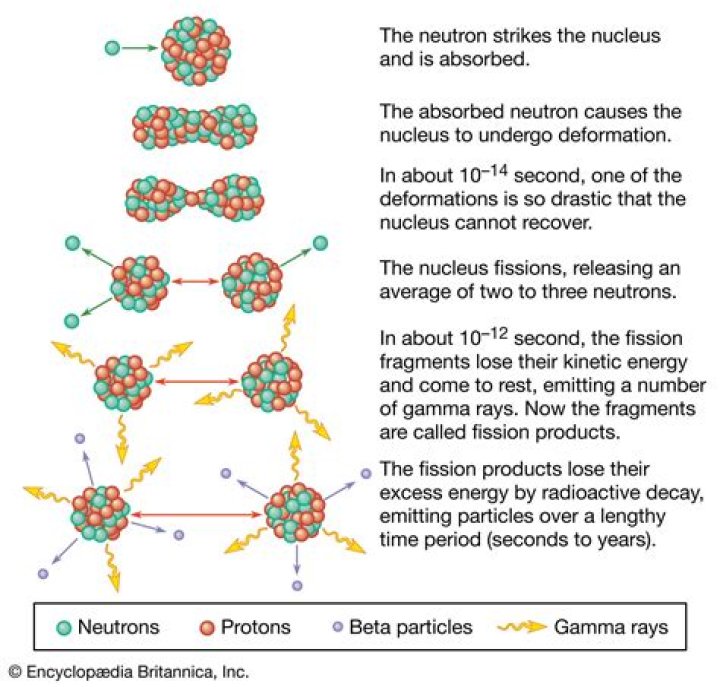
What is fission and fusion?
Fission is the splitting of a heavy, unstable nucleus into two lighter nuclei, and fusion is the process where two light nuclei combine together releasing vast amounts of energy. While different, the two processes have an important role in the past, present and future of energy creation.
Who discovered the proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
Who found electron?
During the 1880s and ’90s scientists searched cathode rays for the carrier of the electrical properties in matter. Their work culminated in the discovery by English physicist J.J. Thomson of the electron in 1897.
Which is atomic number?
The atomic number is the number of protons in the nucleus of an atom. … In a periodic table arranged in order of increasing atomic number, elements having similar chemical properties naturally line up in the same column (group).
Who found uranium?
Uranium was discovered in 1789 by Martin Klaproth, a German chemist, in the mineral called pitchblende. It was named after the planet Uranus, which had been discovered eight years earlier. Uranium was apparently formed in supernovae about 6.6 billion years ago.
Why is uranium used?
Uranium is a very important element because it provides us with nuclear fuel used to generate electricity in nuclear power stations. … Uranium is also used by the military to power nuclear submarines and in nuclear weapons. Depleted uranium is uranium that has much less uranium-235 than natural uranium.