The key difference between electronegativity and polarity is that electronegativity is the tendency of an atom to attract the electrons in a bond towards it, whereas polarity means the separation of the charges.
How does polarity relate to electronegativity?
Electrons in a polar covalent bond are shifted toward the more electronegative atom; thus, the more electronegative atom is the one with the partial negative charge. The greater the difference in electronegativity, the more polarized the electron distribution and the larger the partial charges of the atoms.
Does polarity increase with electronegativity?
In practice, the polarity of a bond is usually estimated rather than calculated. Bond polarity and ionic character increase with an increasing difference in electronegativity. As with bond energies, the electronegativity of an atom depends to some extent on its chemical environment.
Is 0.4 electronegativity polar or nonpolar?
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.How can you tell if an electronegativity is polar or nonpolar?
(If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.
How do you determine the polarity of a bond?
You’ll need to take the difference between the electronegativity value of the two atom. Their bond polarity is determined according to the range it falls in: Nonpolar covalent: Electronegativity difference < 0.5. Polar covalent: Electronegativity difference is 0.5 – 1.6.
Is an electronegativity difference of 0.5 polar?
Electronegativity DifferenceBond Type0–0.4slightly polar covalent0.5–2.1definitely polar covalent>2.1likely ionic
What is the electronegativity P?
The first scale of electronegativity was developed by Linus Pauling and on his scale phosphorus has a value of 2.19 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).What is polar vs nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.
How does polarity affect solubility?Polarity plays a pivotal role in solubility. A polar solute will dissolve in a polar solvent whereas a non-polar solvent will dissolve in a non-polar solvent. If we put a polar solute in a non-polar solvent, it will not dissolve.
Article first time published onHow do you find electronegativity difference?
Subtract the smaller electronegativity from the larger one to find the difference. For example, if we’re looking at the molecule HF, we would subtract the electronegativity of hydrogen (2.1) from fluorine (4.0).
Which molecule is polar?
Polar molecules are those that possess regions of positive and negative charge. Water is an example of a polar material. The type of bonds it has, when coupled with its shape, gives one end of the molecule a slight positive charge (the hydrogen end) and the other a slight negative charge (the oxygen end).
What does polarity mean in physics?
noun. Physics. the property or characteristic that produces unequal physical effects at different points in a body or system, as a magnet or storage battery. the positive or negative state in which a body reacts to a magnetic, electric, or other field.
What is the difference in electronegativity between 0.5 and 2.0 the difference called?
The rule is that when the electronegativity difference is greater than 2.0, the bond is considered ionic. So, let’s review the rules: 1. If the electronegativity difference (usually called ΔEN) is less than 0.5, then the bond is nonpolar covalent.
What is the polarity of water molecules?
Polarity: Although the net charge of a water molecule is zero, water is polar because of its shape. The hydrogen ends of the molecule are positive and the oxygen end is negative. This causes water molecules to attract each other and other polar molecules.
What determines polarity?
The polarity of a bond is determined by a periodic concept called electronegativity. Electronegativity is an expression of an atom’s tendency to attract electrons in a chemical bond. … If the difference is between 0.4 and 1.7, the bond will be polar. If the difference is greater, the bond will have an ionic character.
What does polarity mean in chemistry?
polarity, in chemical bonding, the distribution of electrical charge over the atoms joined by the bond. … In hydrogen chloride, for example, the hydrogen atom is slightly positively charged whereas the chlorine atom is slightly negatively charged.
Is polar hydrophilic or hydrophobic?
Because polar molecules are generally water soluble, they are referred to as being hydrophilic, or water-loving. The one-carbon alcohol, methanol, is an example of a polar molecule.
Is ch4 polar or nonpolar?
Methane (CH4) is a non-polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms. Methane is non-polar as the difference in electronegativities between carbon and hydrogen is not great enough to form a polarized chemical bond.
How do you find the electronegativity of an atom?
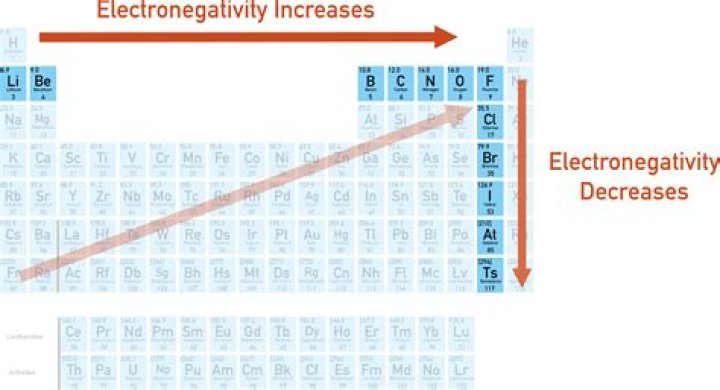
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
How are electronegativity polarity and water solubility related?
Atom with greater electronegativity develops a negative charge and the atom attached to it develops a positive charge. This induced charge affects the solubility of the compounds in solvents. A polar compound gets easily dissolved in polar solvent as polar-polar interactions develop between the two.
How does polarity affect solubility in chromatography?
Polarity has a huge affect on how attracted a chemical is to other substances. … The different colors of pigments have different solubilities based on their polarity. The blue pigment has a greater solubility in water so it moves faster (goes higher) through the chromatography paper.
Why do polar dissolve polar?
Polar solvents will dissolve polar and ionic solutes because of the attraction of the opposite charges on the solvent and solute particles. Non-polar solvents will only dissolve non-polar solutes because they cannot attract the dipoles or the ions.
What is bond polarity?
In chemistry, bond polarity is the separation of electric charge along a bond, leading to a molecule or its chemical groups having an electric dipole or dipole moment. Electrons are not always shared equally between two bonding atoms. … Electronegativity measures a particular atom’s attraction for electrons.
Is it possible to have a polar bond but the molecule polarity is nonpolar?
For the most part, there is a direct correlation between the polarity of a molecule and number and types of polar or non-polar covalent bonds which are present. In a few cases, a molecule may have polar bonds, but in a symmetrical arrangement which then gives rise to a non-polar molecule such as carbon dioxide.
What accounts for the polarity of compounds?
The polarity of a compound depends on how the atoms within the compound are attracted to each other. This attraction can create a difference in electronegativity if one atom or molecule has more “pull” than another and make the molecule polar.
What does nonpolar mean?
Definition of nonpolar : not polar especially : consisting of molecules not having a dipole a nonpolar solvent.
What does polar mean in water?
Water is a “polar” molecule, meaning that there is an uneven distribution of electron density. Water has a partial negative charge ( ) near the oxygen atom due the unshared pairs of electrons, and partial positive charges ( ) near the hydrogen atoms.
What is polarity in biology?
The Oxford Dictionaries definition of polarity for biology is: “the tendency of living organisms or parts to develop with distinct anterior and posterior (or uppermost and lowermost) ends, or to grow or orientate in a particular direction” [1].
What does same polarity mean?
Magnetic polarity refers to the orientation of these poles in space. … If both voltages have the same polarity, the signal is called unipolar; if the voltages have opposite polarity, the signal is called bipolar.
What is called polarity?
Definition of polarity 1 : the quality or condition inherent in a body that exhibits opposite properties or powers in opposite parts or directions or that exhibits contrasted properties or powers in contrasted parts or directions : the condition of having poles.