Electronegativity Of Fluorine
What is the electronegativity difference between calcium and fluorine?
The first electronegativity scale was developed by Linus Pauling, and on the scale, calcium has a value of 1.00 on a scale of 0.7 (an estimate for francs) to 2.20 (for 1 hydrogen) to 3 , 98 (fluorine).
He also asked what is the electronegativity of football?
1Does calcium also have high or low electronegativity?
So the lowest value will be on the lower left - of these atoms, therefore, cesium. Electronegativity increases over time. Electronegativity increases when you join a group. Calcium is higher in the group than barium, so it has higher electronegativity.
Also, is F or C more electronegative?
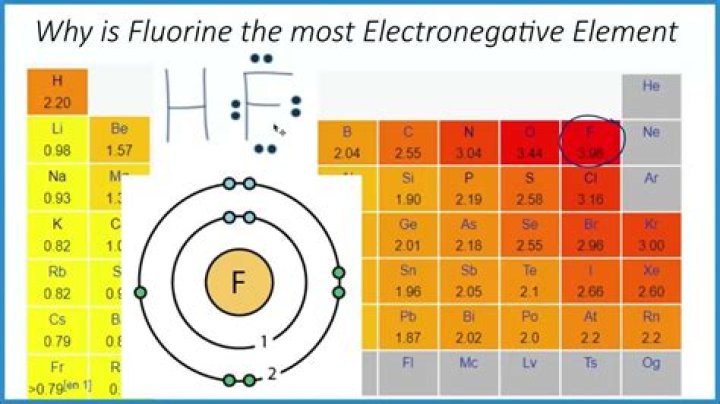
Cesium is the least electronegative element in the periodic table (with a Pauling value of 0.79) while fluorine is the most electronegative element (3.98).
What is the most electronegative O or CL?
Oxygen is more electronegative than chlorine for the following reasons: In an oxygen atom, the nuclear charge of the outermost electrons is high, but slightly lower than that of fluorine, since one electron is only less present than fluorine and therefore the second it is more electronegative. the item.
Where is the electronegativity on the periodic table?
In the periodic table, electronegativity usually increases as you move from left to right over time and decreases as you step down from a group. Consequently, most of the electronegative elements are found in the upper right of the periodic table, while the less electronegative elements are found in the lower left.
Is HCL polar or non-polar?
HCl is a polar molecule because chlorine has a higher electronegativity than hydrogen. It then attracts electrons to pass more time, which gives it a negative charge and hydrogen a positive charge.
How do you know if Br2 is polar or non-polar?
Which group has the highest electronegativity?
Fluorine
Which element has the lowest electronegativity?
Francio
Which element has the highest ionization energy?
This trend shows that cesium has the lowest ionization energy and fluorine has the highest ionization energy (excluding helium and neon).
Why does fluorine have the highest electronegativity?
Electronegativity of fluorine Fluorine is the most electronegative element as it has 5 electrons in its 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons. Because fluorine comes so close to the ideal electronic configuration, the electrons are kept very close to the nucleus.
Which element has the greatest NP electronegativity?
Fluorine Is lubrication on C electronegative?
Phosphorus and sulfur belong to the same time and are conceptually more electronegative than phosphorus. Carbon should be the most electronegative of the three.
What makes something more electronegative?
The electronegativity of an atom is affected both by its atomic number and by the distance between the valence electrons of the charged nucleus. The greater the number of associated electronegativity, the more electrons an atom or substituent group will attract.
Is it more electronegative than N?
Carbon has an electronegativity of 2.55 while nitrogen has an electronegativity of 3.04. Is
CL more electronegative than C?
Cl is relatively larger than carbon and only needs one electron to fill the valence shell and be stable. the charge / mass ratio is quite high. Electronegativity is about 3.5 directly after fluorine. Due to its nuclear charge and size, it cannot process more than 4 electrons in the valence shell.
Is sulfur more electronegative than oxygen?
Sulfur (EN = 2.58) is much less electronegative than oxygen (EN = 3.44). 4. Sulfur can expand the valence shell so that it contains more than eight electrons, but without oxygen.