π bonds are those with one node that is in-plane with the internuclear axis. π bonds can for between two d orbitals or between d orbitals and other types of orbitals with comparable symmetry. … d orbitals can also form π bonds using p orbitals with compatible symmetry, as shown in Figure 5.1. 3.
Can p and d orbitals form pi bonds?
Yes d orbital can form pi bond with p orbital of other participating atom called p(pi) -d(pi) bond. Similarly if it overlapped sidewise with the two lobes d orbital of other participating atom then it is called d(pi)-d(pi) bond.
Which orbital Cannot pi bond?
The orbitals p and d can form π bonds, but the s-orbital cannot form a π bond.
What orbitals make pi bonds?
Pi bonds are formed from the overlap of parallel p orbitals on adjacent atoms. They are not formed from hybrid orbitals.Can dx2 y2 form pi bond?
If dx^2 – y^2 and py orbitals come close together along z – axis, then they can form pi – bond by sideways overlapping.
What orbitals form pi and sigma bonds?
It is important to realize, however, that the two bonds are different: one is a sigma bond, while the other is a pi bond. … The sp hybrid orbitals form a sigma bond between each other as well as sigma bonds to the hydrogen atoms. Both the py and the pz orbitals on each carbon atom form pi bonds between each other.
Which of the following has PΠ − Dπpπ − DΠ bonding?
The correct option is : b SO32– Explanation:In sulphite ion the central atom sulphur is sp3 hybridised. Electronic structure of S atom in excited stateThe three p electrons form o bonds with three oxygen atoms – with one position of the tetrahedron being occupied by a lone pair.
Can sp2 orbitals form pi bonds?
When two sp2 hybridized carbon atoms approach each other to bond, two sp2 orbitals approach each other head to head, and two p orbitals approach each other sideways. The bond formed by the sp2 orbitals is a sigma bond, and the bond formed by the p orbitals is called a pi bond.Why hybrid orbitals do not form pi bonds?
The new orbitals formed are called hybrid orbitals. … While pi bond is not formed by overlapping of hybrid orbitals. They are formed by side wise overlapping of those atomic orbitals which do not undergo hybridisation process.
Which d orbitals can form Delta bond?Delta bonds are formed by side way overlap of four lobes of d-orbitals. For example: the d(xy) of one metal can overlap with d(xy) of other metal.
Article first time published onWhich of the approaching axis is not appropriate to form bond by two py orbitals?
That simply implies that there will be no sidewise overlap between orbitals in that axis, and hitherto there will be no π-bond formation. For the p-orbitals, the orbitals, thus, cannot form any π-bonds. In similar manner, the orbitals which aredirected in the Y-axis cannot form π-bonds.
Can d orbitals form sigma bonds?
d orbitals can also form σ bonds with other types of orbitals with the appropriate symmetry. Examples of orbitals with appropriate symmetry are the s orbital and certain p orbitals on another atom, as shown below in Figure 5.1.
What are the two nodal planes of the dx2 − y2 Orbital?
There are 2 solutions and when they are plotted into xy plane, two symmetric lines emerge, each one at angle of 45 degrees relative to x or y axes. Both solutions do not mention z variable, thus the nodal regions are actually two planes, both parallel to z axis and making angle of 45 degrees relative to x or y axes.
Does PX and DXY overlap?
Yes, they can form bond.
Is PΠ DΠ bond present in h2co3?
H3PO3 is the correct answer.
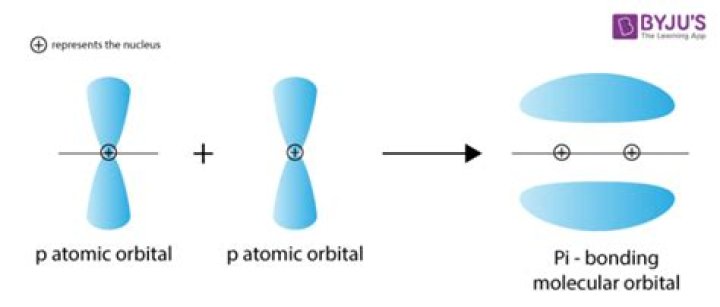
Why do p orbitals form pi bonds?
The sharing of p orbitals creates pi bonds. … π bonds are formed because the side-by-side overlap of p orbitals leads to a lower-energy state than if there were no overlap.
How are sigma and pi bonds formed?
Sigma and pi bonds are formed by atomic orbital overlap. Sigma bonds are formed by the overlapping end – to – end and Pi bonds occur when one atomic orbital lobe overlaps another. … This type of covalent bond is formed by the overlap of bonding orbitals along the internuclear axis from end to end (head-on).
Which hybridizations can form pi bonds?
When two p orbitals overlap side-to-side, they form a π bond. s and p orbitals can form different hybridizations. When two hybridized orbitals overlap, they form a σ bond. sp³-hybridized atoms use all three p orbitals for the hybridization.
What bonds are formed by 4 sp3 hybrid orbitals?
Carbon has four half-filled sp3 hybrid orbitals. Each orbital overlaps with a partially filled 1s atomic orbital of hydrogen to form 4 sigma bonds. To visualize, hydrogen atoms are placed at the four corner of the tetrahedron.
Can sp3d form pi bonds?
The sp3d hybridization gives five relatively equal orbitals. Each of these orbitals can be used to form either sigma bonds or pi bonds depended on the type of atoms it is bonding with in the molecule. So the maximum number of pi bonds that can be formed is five.
How many pi bonds are in SP?
An sp hybridized atom can form two π bonds. An atom uses one s and one p orbital to form two sp hybrid atomic orbitals.
What is D bonding?
In chemistry, delta bonds (δ bonds) are covalent chemical bonds, where four lobes of one involved atomic orbital overlap four lobes of the other involved atomic orbital. … Some rhenium, molybdenum and chromium compounds contain a quadruple bond, consisting of one σ bond, two π bonds and one δ bond.
What are the shapes of d orbitals?
The ml values for five d orbitals are -2, -1, 0, +1, and +2 i.e., we can say d-subshell has five orientations. All these d-orbitals have the same energy and are called degenerate orbitals. The shape of the d-orbitals is given below: Hence, we can say d-orbitals have double dumbbell-shaped.
Which d orbital is having its lobes in between the Axis?
in case of dxy orbital, the four lobes lie in xy plane in between the x and y-axes.
Which of the following orbital Cannot form sigma bond?
s-orbital and s-orbital.
What is true about bonding molecular orbitals?
A bonding molecular orbital is always lower in energy (more stable) than the component atomic orbitals, whereas an antibonding molecular orbital is always higher in energy (less stable).
Why d orbitals do not form Sigma?
Why don’t d orbitals form sigma bonds? – Quora. A sigma bond is formed due to overlapping of singular lobes of two orbitals. A d-orbital has four lobes, so it can form a sigma, pi or even a delta bond. In Dimeric Chromium Acetate, a dz2 orbital overlaps in a sigma fashion to form a bond.
Which combination of atomic orbitals will form σ bonds?
A sigma bond can be formed by overlap of an s atomic orbital with a p atomic orbital. Hydrogen fluoride (HF) is an example: A sigma bond can also be formed by the overlap of two p orbitals.
Which of the following is the nodal plane of D xy orbital?
So in the case of dxy, the nodal planes are the xz and yz planes.
How many nodal planes are there in DXY?
d orbitals have two nodal planes. 6. The number of nodal planes is equal to the value of the angular momentum quantum number, l.
How many nodal planes are there in PX orbital?
Azimuthal quantum number of p-orbital = 1. Therefore, the number of nodal planes in any p-orbital is 1. There are a total 3 p-orbitals and each has 1 nodal plane.