Bh3 Molecular Geometry
What is VSEPR structure and electron geometry for BH3? 3
BH3 is boron trihydride.
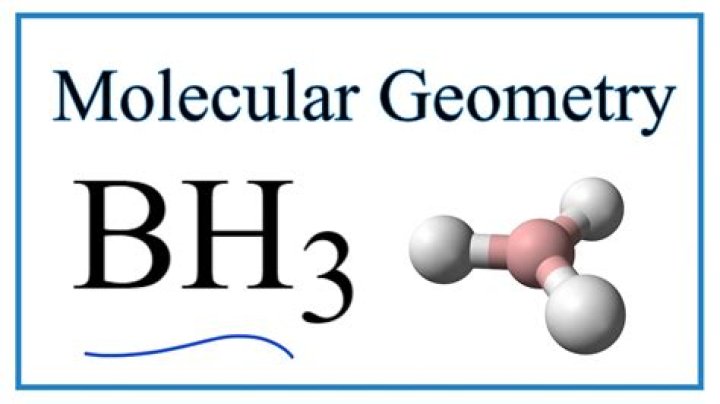
Molecular geometry is a triangular plane.
BH3 is not polar.
That's all I know and I only have to answer the last two questions!
Thank you very much!!!
I checked it first and it looks like Deborah is a dimerk substance.
B2H6, where the hydrogen atom of one of the BH3 pairs forms a detox covalent bond with the other B atom of the dimer. It forms a "curved" shape with two HB links forming 120o at each end and the other HB link forming 97o.
Bh3. Molecular geometry
Because there are no isolated and non-polar couples.
This means that the geometry of the electron is equal to the molecular geometry, i.e. the triangle-planar.
Talk to the IT department.