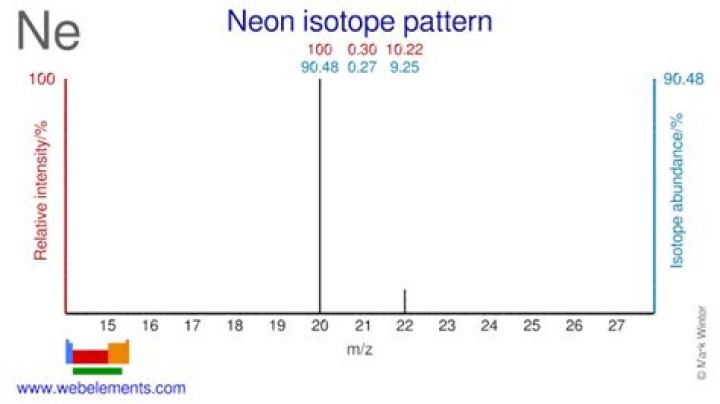
Neon has three isotopes: 20Ne (90.48%), 21Ne (0.27%) and 22Ne (9.25%). Ne and 22Ne are nucleogenic and their variations are well understood. In contrast, 20Ne is not known to be nucleogenic, and the causes of its variation in the Earth have been hotly debated.
How many isotopes are there of neon?
There are three neon isotopes. The more abundant 20Ne and 22Ne are both essentially all primordial, as there is no significant global production of these isotopes.
What are the isotopes of neon used for?
The three Neon isotopes are used for various purposes. Ne-22 is used for the production of the medical radioisotope Na-22. Ne-20 can be used for the production of F-18, although the route via O-18 is by far the most commonly used. Ne-21 has been used in Masers to study quantum physics.
How do you find the isotope of neon?
For neon, which has 10 protons, the mass numbers of the three different naturally occurring isotopes are 20, 21, and 22, corresponding to 10, 11, and 12 neutrons, respectively. Percent means “per hundred.” 90.48% means that 90.48 atoms out of 100 are the isotope with 10 neutrons.What makes neon-22 an isotope?
Aritra G. They are simply isotopes of each other. The Neon – 22 isotope has two extra neutrons in the nucleus than the Neon – 20 isotope. The atomic number and hence the proton number is the same in both.
Is neon an isotope or ion?
NeonDiscovery and first isolationWilliam Ramsay & Morris Travers (1898)Main isotopes of neon
What is the most common isotope for neon?
Isotopeabundancehalf-life (t1/2)20Ne90.48%stable21Ne0.27%stable22Ne9.25%stable
Is neon found in diamonds?
By mass, neon is the 5th most abundant element after carbon, oxygen, helium, and hydrogen. … Ne-20 is stable with 10 neutrons and Ne-21 with 11 neutrons. Elevated Ne-20 abundances have been found in diamonds, and this suggests that there is a solar neon reservoir on the planet.How do you find the atomic weight of neon?
Neon has three naturally occurring isotopes. In a sample of neon, 90.92% of the atoms are Ne-20, which is an isotope of neon with 10 neutrons and a mass of 19.99amu. Another 0.3% of the atoms are Ne-21, which is an isotope of neon with 11 neutrons and a mass of 20.99amu.
What is neon made out of?Neon is created in large mass stars when the internal pressure of the star is great enough to fuse carbon atoms into neon atoms, according to Berkeley Lab. … Neon, along with helium, argon, krypton and xenon, make up the group known as noble gases.
Article first time published onIs neon a non metal?
NameSymbolsMetals/Non-metals/Noble gases/Metalloids(d) MercuryHgMetal(e) FluorineFNon-metal(f) NeonNeNoble Gas(g) SulphurSNon-metal
What is the natural isotopes for neon?
Neon, the second-lightest noble gas, has three stable isotopes and no long-lived radioactive isotopes. The stable isotopes, neon-20, neon-21, and neon-22, are present in a ratio of 9048 : 27 : 925 in the atmosphere and in varying compositions in other materials.
What ions does neon form?
The ions, Ne+, (NeAr)+, (NeH)+, and (HeNe+) are known from optical and mass spectrometric studies. Neon also forms an unstable hydrate.
What is the difference between neon-20 and neon-22?
The only difference between an atom of neon-20 and an atom of neon-22 is the number of neutrons in their nuclei.
Is neon magnetic or nonmagnetic?
GeneralMagnetic orderingnonmagneticThermal conductivity(300 K) 49.1 mW/(m·K)Speed of sound(gas, 0 °C) 435 m/sCAS registry number7440-01-9
Is neon a synthetic element?
There are seven noble gases: helium, neon, argon, krypton, xenon, radon, and oganesson (a synthetic element). … That non-reactivity is what makes neon so useful in light bulbs.
Is neon an ionic compound?
And atoms of neon won’t form covalent structures and share electrons because they already have a full outer shell. So the statement that explains why atoms of neon do not form ionic bonds is that atoms of neon already have a stable octet in their outer shell.
Is neon ionic or covalent?
The extremely stable noble gasses, including helium, neon, argon, krypton, xenon and radon, are all also nonmetal covalent elements. These elements form bonds with one another by sharing electrons to form compounds.
Is neon atomic or molecular?
Furthermore, the curiosity of a stable molecule whose constituent atomic symbols spell the name of one such atom give this structure a unique place in the chemical imagination. Hence, “neon” is both a molecule and an atom.
What is the density of neon?
atomic number10atomic weight20.183melting point−248.67 °C (−415.5 °F)boiling point−246.048 °C (−411 °F)density (1 atm, 0° C)0.89990 g/litre
Why is neon called neon?
In 1898, William Ramsay and Morris Travers at University College London isolated krypton gas by evaporating liquid argon. They had been expecting to find a lighter gas which would fit a niche above argon in the periodic table of the elements. … Ramsay named the new gas neon, basing it on neos, the Greek word for new.
What is the texture of neon?
Classification:Neon is a noble gas and a nonmetalColor:colorlessAtomic weight:20.180State:gasMelting point:-248.57 oC, 24.53 K
How many energy shells does neon have?
Because neon has two atomic shells, it needs two electrons in the first and eight to fill the second. Neon has a total of ten electrons which means two filled shells.
How many energy levels are in neon?
A: A neon atom has two electrons in energy level I and its remaining eight electrons in energy level II, which can hold only eight electrons. This means that is outermost energy level is full. Therefore, a neon atom is very stable.
What is the atomic structure of neon?
Neon is an atom with atomic number ten. Its atomic weight is 20.179 which cause it to have ten neutrons and ten protons in its nucleus and ten electrons outside. Neon; Neon, Ne, is a colorless inert noble gas and it is also the second lightest noble gas.
How do you find the atomic mass of isotopes?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
What is the average atomic mass for all of the isotopes of neon?
(a) The average atomic mass of naturally occurring neon is 20.18 amu. There are two common isotopes of naturally occurring neon as indicated in the table below. (i) Using the information above, calculate the percent abundance of each isotope. Let x represent the natural abundance of Ne-20.
How many isotopes does f have?
Fluorine (9F) has 18 known isotopes ranging from 13F to 31F (with the exception of 30F) and two isomers (18mF and 26mF). Only fluorine-19 is stable and naturally occurring; therefore, fluorine is a monoisotopic and mononuclidic element, and only artificially produced fluorine isotopes have mass numbers other than 19.
How was neon discovered?
Neon was discovered in London in 1898 by a pair of British chemists: Sir William Ramsay and Morris W. … Ramsay chilled a sample of air until it became a liquid. He then warmed this liquid, capturing the gases as they boiled off. Nitrogen, oxygen, and argon had already been identified.
How do you make neon gas?
Neon can be obtained from air by fractional distillation. The first step in fractional distillation of air is to change a container of air to a liquid. The liquid air is then allowed to warm up. As the air warms, each element in air changes from a liquid back to a gas at a different temperature.
Is Neon brittle or malleable?
Neon as a gas in its natural state – completely malleable.